- When should I do the Pap smear test after delivery?
Ideally you should have a medical check up with your doctor 6 – 8 weeks after delivery. During this visit, a general examination including your blood pressure, abdominal and pelvic examination will be done. Pap smear test is also performed at the same setting. In addition, other relevant issues such as contraception are discussed.
- What is a Pap smear?
This is a simple test done by your doctor to have a look at the cells at the neck of your cervix. It is screening test to detect early changes associated with cervical cancer.
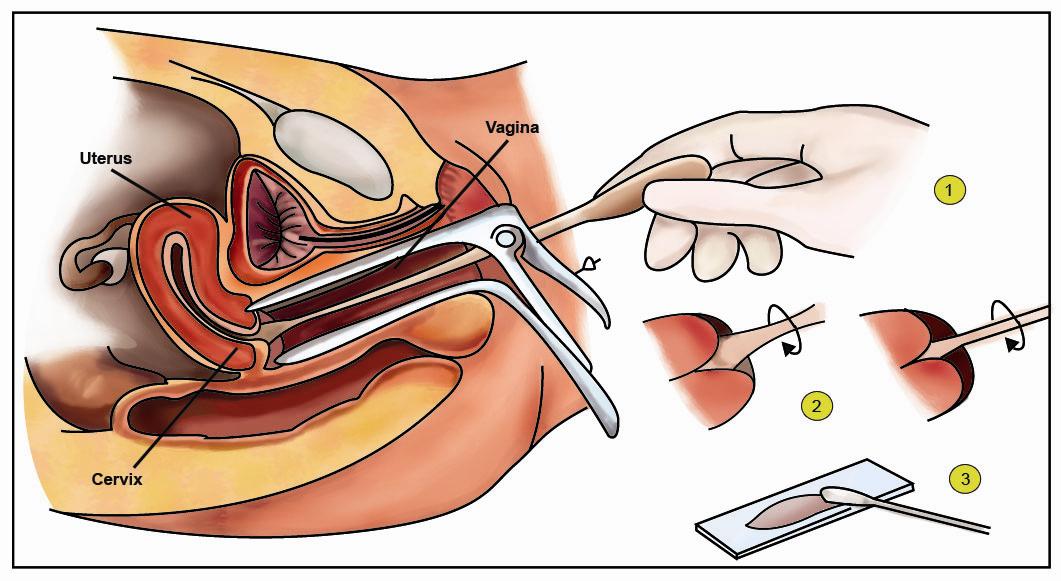
- How is the Pap smear test done?
You would be asked to lie down on a bed, a speculum (a small device to allow your doctor to see the upper part of your vagina and cervix) will be inserted into your vagina. Then, some sample of the cells lining your cervix is taken by scraping the cervix using a plastic brush or wooden spatula. Majority of the women have minimal or no discomfort at all during this test.
- How quickly could I get the result of my Pap smear test back?
Usually it takes about 1 week or more to get the result of your Pap Smear. This will depend on the clinic or hospital you went to.
- What will happen if my Pap Smear test result is abnormal?
This depends on what abnormalities did the Pap Smear test report. Your results may report one of the following:
|
Results
|
Interpretation
|
|
Normal
|
Only normal cells were seen. No precancerous cells were detected.
|
|
Squamous intraepithelial lesion:
i) LSIL (Low grade SIL)
ii) HGSIL (High grade SIL)
|
Some precancerous changes were seen in the cells
Early, mild changes were seen in the shape or size of the cells
Moderate or severe changes were seen in the shape or size of the cells. There is increase risk of ‘precancer’ cells present compared to the LSIL
|
|
ASCUS (Atypical squamous cells)
|
Some abnormal cells were seen. It could be caused by infection, irritation or even precancerous cells.
|
|
AGUS (Atypical glandular cells)
|
Cells changes were seen and require further evaluation
|
|
Cancer
|
There is detection of cancerous cells. You will need further evaluation to detect the spread of cancer.
|
If you have a result which is abnormal, your doctor will suggest several recommendations depending on the results and your risk factors including:
- A repeat pap smear in 3 – 6 months time
- Colposcopy: this is a test that could be done in your doctor’s office whereby your cervix is examined using a microscopy (a magnifying device) If any abnormalities are seen, a small biopsy (small sample of tissue) is removed from the cervix and sent to the lab for further evaluation)
- HPV DNA test: this is a test to detect high risk HPV (human papilloma virus) that is associated with cervical cancer. It uses the Liquid Based Cytology (LBC) technique during their Pap smear test.
- What is Liquid Based Cytology (LBC)?
LBC is a new way of preparing samples taken from the cervix for examination in the laboratory. The sample is collected in the same way as normal conventional Pap Smear. However, instead of smearing the sample onto a glass slide, the brush used to take the smear is placed into a small plastic container containing preservative fluid. This is then sent to the laboratory, spun and treated to remove any pus or mucus. The remaining cells are then taken and smeared into a thin layer of cell on the slides to be inspected under the microscope.
LBC is more expensive than the conventional Pap Smear. However, LBC usage is said to be associated with lower rates of unsatisfactory Pap Smear which then requires repeat samples to be taken, reduced laboratory time to screen the Pap Smear, an increased in detection rates of cancer and reduction in false negative results. However, further studies are needed to confirm the superiority of LBC against conventional Pap Smear test.
- What is the cause of cervical cancer?
More than 95% of cervical cancer is thought to be due by HPV (human papilloma virus) This virus spreads through sexual contact. Approximately 50 – 75% of sexually active women have HPV at some point in their life. There are many different types of HPV. High risk HPV types such as HPV type 16 and 18 causes about 70% of cervical cancer.
- Who is at higher risk of developing cervical cancer?
- Women who had early sexual intercourse < 18 years old
- Women with multiple sexual partners
- Women with high risk HPV infection
- Smokers
- Immunosuppressed
- Is there a way of preventing or reducing the risk of getting cervical cancer?
- Having regular Pap smear test. This would detect early precancerous cell changes that occur. Hence, earlier referral and treatment could be done before cancer develops.
- HPV vaccination. Currently, you could be vaccinated against certain types high risk HPV which is commonly associated with cervical doctor. There are 2 types of vaccine available currently i.e Cervarix and Gardasil. You could ask your doctor about it.
- If I have taken my cervical cancer vaccination, could I stop taking my Pap smear test?
No, you should continue doing you Pap smear test as instructed by your doctor.
- How often should I do my Pap smear test?
The recommendation depends on your age and risk factors. In general, it is recommended to have yearly Pap smear test. If you have 3 consecutively normal yearly Pap smear result, you could do 2 – 3 yearly Pap smear. However, if you have previous abnormal results or risk factors your doctor may ask you to do it more frequently.
REFERENCES
Richard Todd, Mahmood Shafi. Invasive cervical cancer. Current Obstetrics and Gynaecology 2004; 14: 200 -206
Cheung AN, Szeto ET, Leung BS, et al. Liquid-based cytology and conventional cervical smears: a comparison in an Asian screening population. Cancer 2003; 99:331 – 5
Abulafia O, Pezzullo JC, Sherer DM. Performance of ThinPrep liquid-based cervical cytology in comparison with conventionally prepared Papanicolaou smear: a quantitative survey. Gynecol Oncol 2003; 80:137-44
Renshaw AA, Young NA, Birdsong GG, et al. Comparison of performance of conventional and ThinPrep gynecologic preparations in the College of American Pathologists Gynaecologic Cytology Program. Arch Pathol Lab Med 2004; 128:17-22
Filed under Post Partum Care | No Comments








